
Other, more exotic states of matter can occur at extremely high energy levels or at extremely low temperatures, where atoms and molecules (or their components) arrange in unusual ways. Plasmas occur naturally in flames, lightning and auroras. 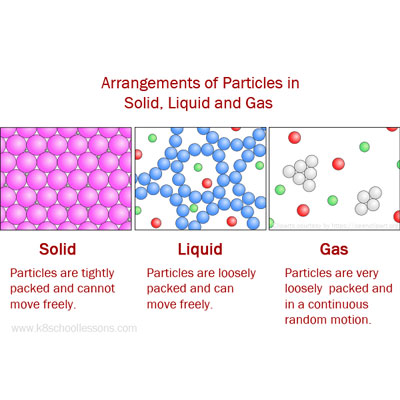
This means that a plasma has very different properties from those of an ordinary gas. While it’s similar to a gas the electrons are free in a cloud rather than attached to individual atoms. Plasma is sometimes referred to as a fourth state of matter. The atoms and molecules move freely and spread apart from one another. They move around but stay close together. In a liquid, the atoms and molecules are loosely bonded. Liquids – definite volume but able to change shape by flowing.  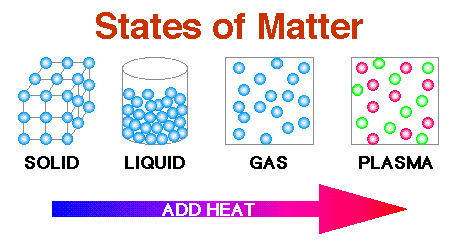
They vibrate in place but don’t move around. In a solid, the atoms and molecules are attached to each other.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
